What Are Factors That May Affect Breast Density?
1. Can breast density be changed with diet? Is fat in the diet related to breast density?
Dietary fat intake has little to do with breast density; however, it does relate to increased body mass index (BMI). BMI is a measure of body fat based on height and weight, and there is more fatty breast tissue in women with higher BMI. Higher BMI reduces the percent of density but might not reduce the total amount of dense tissue. BMI and breast density are both separate risk factors for breast cancer. Before menopause, low BMI [1, 2] increases the risk of breast cancer. After menopause, weight gain and increasing BMI increase the risk of breast cancer.
References Cited
1. van den Brandt PA, Spiegelman D, Yaun SS, et al. Pooled analysis of prospective cohort studies on height, weight, and breast cancer risk. Am J Epidemiol 2000; 152:514-527
2. Huo CW, Chew GL, Britt KL, et al. Mammographic density-a review on the current understanding of its association with breast cancer. Breast Cancer Res Treat 2014; 144:479-502
2. Can exercise affect a woman’s breast density category?
It can. While exercise can decrease the amount of fat in the breast, the glandular or dense breast tissue is not affected by exercise. So, if a woman loses a lot of weight due to exercise, her breasts can appear more dense due to loss of fat (the amount of fat decreases while the amount of dense tissue remains the same) [1].
While exercise does not decrease the actual amount of dense tissue in the breasts, it does decrease the overall risk of developing breast cancer [2].
References Cited
1. Peters TM, Ekelund U, Leitzmann M, et al. Physical activity and mammographic breast density in the EPIC-Norfolk cohort study. Am J Epidemiol 2008; 167:579-585
2. Azam S, Kemp Jacobsen K, Aro AR, et al. Regular physical activity and mammographic density: a cohort study. Cancer Causes & Control 2018; 29:1015-1025
3. Does taking hormone replacement therapy (HRT) or estrogen therapy alone affect breast density or breast cancer risk?
- Increases in both breast density [1, 2] and breast cancer risk [3, 4] have been shown in patients taking oral or transdermal combined estrogen and progesterone hormone supplements (i.e. hormone replacement therapy, HRT).
- In women who have had breast cancer, oral or transdermal HRT have been shown to increase risk of breast cancer recurrence [5].
- Data are conflicting as to whether taking oral or transdermal estrogen alone in women who have had a hysterectomy increases breast density [5-9] or breast cancer risk [3, 4].
- There are minimal data on the effect of vaginal estrogen on breast density.
- In women who have not had breast cancer, vaginal estrogen has not been shown to increase breast cancer risk.
- In women who have had breast cancer, vaginal estrogen has not been shown to increase breast cancer recurrence, except in one study that reported an increased risk in breast cancer survivors who took aromatase inhibitors [6]. Vaginal estrogen has not been shown to increase death from breast cancer in breast cancer survivors [6, 13].
A large meta-analysis of prospective observational studies found that ever users of HRT had a 30% greater risk of developing breast cancer than never users [relative risk (RR) 1.29 (95% CI 1.27, 1.30)][3]. Risk increased with duration of use for both current and past users, with higher risk at any given duration of use for current users [current users, RR 1.20 (95% CI 1.01-1.43) for <1 year of use to 2.51 (95% CI 2.35-2.68) for ≥ 15 years of use; past users, RR 1.02 (95% CI 0.95-1.10) for <1 year of use to 1.30 (95% CI 1.25-1.37) for ≥ 15 years of use]. Similarly, the Women’s Health Initiative randomized trial found nearly 30% greater risk after a median 20 years of follow-up among women randomized to estrogen and medroxyprogesterone acetate (hazard ratio 1.28; 95% CI 1.13-1.45) [4].
Data are conflicting as to whether taking oral or transdermal estrogen alone in women who have had a hysterectomy increases breast density [7-11] or breast cancer risk [3, 4]. There are minimal data on the effect of vaginal estrogen on breast density. Vaginal estrogen has not been shown to increase breast cancer risk [3, 12] in women who have not had breast cancer.
In women who have had breast cancer, oral or transdermal HRT have been shown to increase risk of breast cancer recurrence [5]. Vaginal estrogen has not been shown to increase risk of breast cancer recurrence, except in one study that reported an increased risk in breast cancer survivors who used vaginal estrogen and took aromatase inhibitors [6]. Vaginal estrogen has not been shown to increase death from breast cancer in women who have had breast cancer [6, 13].
References Cited
1. Azam S, Jacobsen KK, Aro AR, Lynge E, Andersen ZJ. Hormone replacement therapy and mammographic density: a systematic literature review. Breast Cancer Res Treat 2020;182(3):555-579.
2. McTiernan A, Martin CF, Peck JD, et al.; Women’s Health Initiative Mammogram Density Study Investigators. Estrogen-plus-progestin use and mammographic density in postmenopausal women: Women’s Health Initiative randomized trial. J Natl Cancer Inst 2005;97(18):1366-76.
3. Collaborative Group on Hormonal Factors in Breast Cancer. Type and timing of menopausal hormone therapy and breast cancer risk: individual participant meta-analysis of the worldwide epidemiological evidence. Lancet 2019 28;394(10204):1159-1168.
4. Chlebowski RT, Anderson GL, Aragaki AK, et al. Association of Menopausal Hormone Therapy With Breast Cancer Incidence and Mortality During Long-term Follow-up of the Women’s Health Initiative Randomized Clinical Trials. JAMA 2020;324(4):369-380.
5. Poggio F, Del Mastro L, Bruzzone M, et al. Safety of systemic hormone replacement therapy in breast cancer survivors: a systematic review and meta-analysis. Breast Cancer Res Treat 2022;191(2):269-275.
6. Cold S, Cold F, Jensen MB, Cronin-Fenton D, Christiansen P, Ejlertsen B. Systemic or Vaginal Hormone Therapy After Early Breast Cancer: A Danish Observational Cohort Study. J Natl Cancer Inst 2022;114(10):1347-1354.
7. Greendale GA, Reboussin BA, Slone S, Wasilauskas C, Pike MC, Ursin G. Postmenopausal hormone therapy and change in mammographic density. J Natl Cancer Inst 2003;95(1):30-7.
8. Grady D, Vittinghoff E, Lin F, et al. Effect of ultra-low-dose transdermal estradiol on breast density in postmenopausal women. Menopause 2007;14:391-396.
9. Maskarinec G, Pagano I, Lurie G, Kolonel LN. A longitudinal investigation of mammographic density: the multiethnic cohort. Cancer Epidemiol Biomarkers Prev. 2006;15(4):732-9.
10. McTiernan A, Chlebowski RT, Martin C, et al. Conjugated equine estrogen influence on mammographic density in postmenopausal women in a substudy of the women’s health initiative randomized trial. J Clin Oncol 2009;20;27(36):6135-43.
11. Aiello EJ, Buist DS, White E. Do breast cancer risk factors modify the association between hormone therapy and mammographic breast density? (United States). Cancer Causes Control 2006;17(10):1227-35.
12. Crandall CJ, Hovey KM, Andrews CA, et al. Breast cancer, endometrial cancer, and cardiovascular events in participants who used vaginal estrogen in the Women’s Health Initiative Observational Study. Menopause 2018 Jan;25(1):11-20.
13. McVicker L, Labeit AM, Coupland CAC, et al. Vaginal Estrogen Therapy Use and Survival in Females With Breast Cancer. JAMA Oncol 2024;10(1):103-108.
4. Is there anything a patient can do to decrease her breast density? What about taking tamoxifen?
Tamoxifen blocks the estrogen receptor in breast cells and in breast cancer cells which express the estrogen receptor. Tamoxifen may be recommended to reduce the risk of developing breast cancer in women who have had prior atypical biopsies. Tamoxifen is also prescribed for women who have had breast cancer that expresses estrogen receptors to decrease recurrence. One study [1] found that when breast density is carefully measured by computer software, women whose breasts became at least 10% less dense while taking tamoxifen had a 63% reduction in risk of developing breast cancer – whereas those whose breast density did not change did not see a decrease in their risk. Several similar studies [2, 3] in women who have had breast cancer showed that only women whose breast density decreased on tamoxifen had decreased risk of recurrence. Tamoxifen also carries about a 3% risk of blood clots (which could result in pulmonary embolism or stroke) and a smaller risk of endometrial cancer (if the woman still has her uterus).
If a woman is on hormone therapy for menopausal symptoms, her breast density may decrease if she stops taking hormone supplements.
References Cited
1. Cuzick J. Breast density predicts endocrine treatment outcome in the adjuvant setting. Breast Cancer Res 2012; 14:109
2. Cuzick J, Warwick J, Pinney E, et al. Tamoxifen-induced reduction in mammographic density and breast cancer risk reduction: a nested case-control study. J Natl Cancer Inst 2011; 103:744-752
3. Li J, Humphreys K, Eriksson L, Edgren G, Czene K, Hall P. Mammographic density reduction is a prognostic marker of response to adjuvant tamoxifen therapy in postmenopausal patients with breast cancer. J Clin Oncol 2013; 31:2249-2256
5. Will taking Arimidex or other aromatase inhibitors affect breast density?
Aromatase inhibitors block the body’s own production of estrogen and are prescribed for postmenopausal women who have had breast cancer where the tumor cells express receptors for estrogen. One study [1, 2] looked at women who have had breast cancer. When breast density is carefully measured by computer software, women who experienced a decrease in breast density while taking tamoxifen or aromatase inhibitors had a lower risk of recurrence than women who did not experience a decrease in breast density.
References Cited
1. Kim J, Han W, Moon HG, et al. Breast density change as a predictive surrogate for response to adjuvant endocrine therapy in hormone receptor positive breast cancer. Breast Cancer Res 2012; 14:R102
2. Kim J, Han W, Moon HG, et al. Erratum to Breast density change as a predictive surrogate for response to adjuvant endocrine therapy in hormone receptor positive breast cancer. Breast Cancer Res 2012; 14:403
6. For breast cancer survivors, is there a correlation between dense breasts and the likelihood of cancer in the opposite breast?
Yes. There is a 1.8-fold higher risk of cancer in the opposite breast among women with dense breasts [1], but density and associated risk can be reduced with treatment. A 10% decrease of mammographic density or more within the first two years after an original diagnosis, as a result of treatment, is associated with a significantly reduced risk of cancer in the opposite breast (known as contralateral breast cancer) [2]. This potential new risk predictor can thus contribute to decision-making in follow-up treatment – particularly the continuation of a chemoprevention drug, like tamoxifen or aromatase inhibitors, which reduce breast density in some women [3].
References Cited
1. Raghavendra A, Sinha AK, Le-Petross HT, et al. Mammographic breast density is associated with the development of contralateral breast cancer. Cancer 2017; 123:1935-1940
2. Sandberg ME, Li J, Hall P, et al. Change of mammographic density predicts the risk of contralateral breast cancer: a case-control study. Breast Cancer Res 2013; 15:R57
3. Engmann NJ, Scott CG, Jensen MR, et al. Longitudinal changes in volumetric breast density with Tamoxifen and aromatase inhibitors. Cancer Epidemiol Biomarkers Prev 2017; 26:930-937
7. Is breast density an issue that affects men?
Not normally, though rarely men do get breast cancer. Normal male breasts are mostly fatty. Sometimes men’s breasts do become enlarged and develop glandular tissue due to a condition called gynecomastia. This enlargement, due to a hormonal imbalance, normally affects one breast more than the other. If a mammogram is performed, it is usually easy to distinguish gynecomastia from breast cancer.
8. Is breast density inherited?
Breast density is at least partially inherited, though it is complex to predict. If a patient’s mother had dense breast tissue, it is more likely the patient will too.
9. Does the increase in density due to breastfeeding affect the ability of screening tests, like a mammogram, to find breast cancer?
Yes. Changes in the breast during breastfeeding do reduce the accuracy of screening tests (like mammography or MRI). Unless the patient plans to be breastfeeding for more than one to two years and is at high risk, it is generally recommended to wait at least a few months after breastfeeding stops before resuming breast screening. Ultrasound is usually performed first if a patient is breastfeeding and there is a concern about breast symptoms. If a breastfeeding woman needs to undergo breast imaging, it is advisable to nurse or pump immediately before the exam.
10. Do Black women have denser breasts? Do Asian women have denser breasts? Do Hispanic women have denser breasts?
Analysis of over 2.6 million mammogram results from the Breast Cancer Surveillance Consortium confirms greater prevalence of dense breasts in Asian women (66%) than non-Hispanic (NH) White women (45%), Hispanic women (45%), or Black women (37%). Breast density is inversely related to body mass index (BMI) and decreases with age and menopause. After correcting for BMI, age, and menopausal status, Black women were more likely to have dense breasts than NH White or Hispanic women.
References Cited
1. Kerlikowske K, Bissell MCS, Sprague BL, et al. Impact of BMI on Prevalence of Dense Breasts by Race and Ethnicity. Cancer Epidemiol Biomarkers Prev 2023:OF1-OF7
2. del Carmen MG, Halpern EF, Kopans DB, et al. Mammographic breast density and race. AJR Am J Roentgenol 2007; 188:1147-1150
3. McCarthy AM, Keller BM, Pantalone LM, et al. Racial differences in quantitative measures of area and volumetric breast density. J Natl Cancer Inst 2016; 108
11. Are there any special considerations for screening of Black or Hispanic women?
Black women are more likely to have disease-causing variants (mutations) in BRCA. The American College of Radiology recommends Black women be evaluated in a high-risk program by age 30, even in the absence of family history [1]. Further, it is more important for Black women and Hispanic women to start screening at least by age 40 as peak breast cancer incidence occurs in the mid 40s, compared to peak in the early 50s for Caucasian women [2].
References Cited
1. Monticciolo DL, Newell MS, Moy L, Niell B, Monsees B, Sickles EA. Breast cancer screening in women at higher-than-average risk: Recommendations from the ACR. J Am Coll Radiol 2018; 15:408-414
2. Stapleton SM, Oseni TO, Bababekov YJ, Hung YC, Chang DC. Race/ethnicity and age distribution of breast cancer diagnosis in the United States. JAMA Surg 2018; 153:594-595
12. Is there a relationship between having dense breasts and pathogenic BRCA gene variants (mutations)?
Disease-causing BRCA gene variants and dense breasts are each independent risk factors for the development of breast cancer; however, pathogenic BRCA1 or BRCA2 variants are associated with a much higher risk than that of having dense breasts. As BRCA1 and BRCA2 disease-causing variants are associated with a higher risk, MRI is part of routine screening beginning at age 25 to 30 for women who have these variants, regardless of breast density. Cancers are also more likely to develop at a younger age in women with disease-causing variants in BRCA genes, and the breasts are usually dense at younger ages making mammography especially ineffective as a standalone test in such women.
13. What is the recommended breast screening for women with disease-causing genetic variants (mutations)?
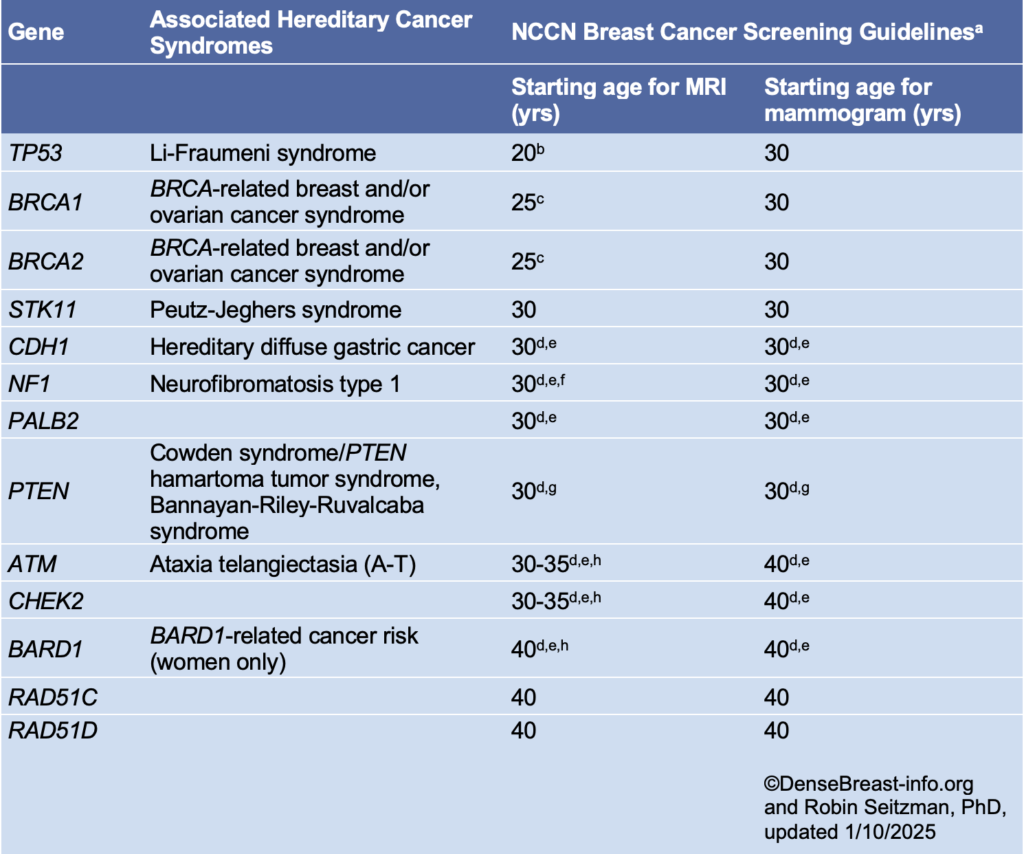
National Comprehensive Cancer Network (NCCN) guidelines recommend annual screening MRI beginning by age 25, with the addition of mammography beginning at age 30, in women who are known to carry pathogenic/likely pathogenic variants in BRCA1 or BRCA2 (unless the woman has had bilateral mastectomy), and in women who are untested first-degree relatives of those with known pathogenic/likely pathogenic variants (see table below) [1].
Women who are known to carry or are first-degree untested relatives of individuals with less common disease-causing variants [such as those associated with Li-Fraumeni syndrome (TP53); Bannayan-Riley-Ruvalcaba syndrome or Cowden syndrome (PTEN); hereditary diffuse gastric cancer (CDH1); Peutz-Jeghers syndrome (STK11); Neurofibromatosis type 1 (NF1); or Fanconi anemia (PALB2)] are also recommended for annual screening MRI, with age to start depending on the variant (see table below). Women with NF1 have elevated risk only through age 50: supplemental MRI should stop beyond age 50 for these women. For women with disease-causing variants in genes other than NF1, supplemental MRI screening should continue until age 75, after which screening should continue on an individual basis [1].
In addition to pathogenic/likely pathogenic variant carriers and their untested first-degree relatives, annual screening MRI is recommended in addition to mammography/tomosynthesis in other subgroups of women as detailed in the Dense Breasts/High-Risk Screening Guidelines.
NCCN Breast Cancer Screening Guidelines in Women Who Carry or Are First-Degree Untested Relatives of Individuals with Pathogenic Variants or Variants Known or Likely to Increase Breast Cancer Risk [1]*
*Please refer to the NCCN guideline website for updates.
a Breast awareness starting at age 18 years. Clinical breast exam every 6-12 months starting at age 25 years or 5-10 years before the earliest known breast cancer in the family. Screening should continue as indicated until age 75 (other than for women with a pathogenic NF1 mutation), after which screening should be considered on an individual basis [1].
b Start at age 20 or the age of the earliest diagnosed breast cancer in the family if younger than age 20. Adding mammography at age 30 is preferred due to the potential risk from radiation exposure in pathogenic/likely pathogenic variant carriers under age 30. Mammography can be performed from ages 20-29 years only if MRI is unavailable.
c Begin at age 25, or starting age may be individualized based on family history of a breast cancer diagnosis before at 30. From age 25-29 years, breast MRI alone is recommended. Adding mammography at age 30 is preferred due to the potential risk of radiation exposure in pathogenic/likely pathogenic variant carriers under age 30. Mammography can be performed from ages 25-29 years only if MRI is unavailable.
d For women with pathogenic/likely pathogenic variants who are treated for breast cancer and have not had bilateral mastectomy, screening should continue as described.
e Start at stated age or 5-10 years before the earliest known breast cancer in the family (whichever comes first), or based on specific pathogenic/likely pathogenic variant.
f There are currently no data to suggest an increased breast cancer risk after age 50 years in women with NF1; therefore, MRI screening may discontinue at 50 years of age in this group. In addition, the presence of breast neurofibromas may lead to false-positive MRI results; more data on sensitivity and specificity of MRI in women with NF1 are needed.
g Start at stated age or 10 years before the earliest known breast cancer in the family (whichever comes first).
h The use of MRI in these patients depends on a number of risk factors, including family history, age, breast density, and patient preference.
References Cited
1. National Comprehensive Cancer Network. Genetic/familial high-risk assessment: Breast, ovarian, pancreatic, and prostate. (Version 2.2025). Available at https://www.nccn.org/professionals/physician_gls/pdf/genetics_bopp.pdf.
14. How do women under the age of 40 find information about breast density and their risks for breast cancer?
Dense breasts are mostly an issue affecting mammography performance so a patient generally does not need to know until they begin having mammograms. For women at normal risk, mammography is often recommended to begin at age 40. If a woman has a family history of breast cancer and has not begun mammography screening, she should speak to her doctor about personal risk factors and when mammography and possibly other screening should begin. As a general guide, if a woman’s mother or sister was diagnosed with breast cancer before age 50, she may want to begin annual screening 10 years before the relative’s age at diagnosis, but not before age 30.
European guidelines recommend double-read biennial screening digital mammography for average-risk women 50 to 69 years of age. Also encouraged are biennial screening for women 73 to 75 years of age, and annual screening for ages 40–45 to 49 [1].
References Cited
1. Sardanelli F, Aase HS, Alvarez M, et al. Position paper on screening for breast cancer by the European Society of Breast Imaging (EUSOBI) and 30 national breast radiology bodies from Austria, Belgium, Bosnia and Herzegovina, Bulgaria, Croatia, Czech Republic, Denmark, Estonia, Finland, France, Germany, Greece, Hungary, Iceland, Ireland, Italy, Israel, Lithuania, Moldova, The Netherlands, Norway, Poland, Portugal, Romania, Serbia, Slovakia, Spain, Sweden, Switzerland and Turkey. Eur Radiol 2017; 27:2737-2743