Yes. There is a 1.8-fold higher risk of cancer in the opposite breast among women with dense breasts [1], but density and associated risk can be reduced with treatment. A 10% decrease of mammographic density or more within the first two years after an original diagnosis, as a result of treatment, is associated with a significantly reduced risk of cancer in the opposite breast (known as contralateral breast cancer) [2]. This potential new risk predictor can thus contribute to decision-making in follow-up treatment – particularly the continuation of a chemoprevention drug, like tamoxifen or aromatase inhibitors, which reduce breast density in some women [3].
E.U.
Is breast density an issue that affects men?
Not normally, though rarely men do get breast cancer. Normal male breasts are mostly fatty. Sometimes men’s breasts do become enlarged and develop glandular tissue due to a condition called gynecomastia. This enlargement, due to a hormonal imbalance, normally affects one breast more than the other. If a mammogram is performed, it is usually easy to distinguish gynecomastia from breast cancer.
Is breast density inherited?
Breast density is at least partially inherited, though it is complex to predict. If a patient’s mother had dense breast tissue, it is more likely the patient will too.
Does the increase in density due to breastfeeding affect the ability of screening tests, like a mammogram, to find breast cancer?
Yes. Changes in the breast during breastfeeding do reduce the accuracy of screening tests (like mammography or MRI). Unless the patient plans to be breastfeeding for more than one to two years and is at high risk, it is generally recommended to wait at least a few months after breastfeeding stops before resuming breast screening. Ultrasound is usually performed first if a patient is breastfeeding and there is a concern about breast symptoms. If a breastfeeding woman needs to undergo breast imaging, it is advisable to nurse or pump immediately before the exam.
Do Black women have denser breasts? Do Asian women have denser breasts? Do Hispanic women have denser breasts?
Analysis of over 2.6 million mammogram results from the Breast Cancer Surveillance Consortium confirms greater prevalence of dense breasts in Asian women (66%) than non-Hispanic (NH) White women (45%), Hispanic women (45%), or Black women (37%). Breast density is inversely related to body mass index (BMI) and decreases with age and menopause. After correcting for BMI, age, and menopausal status, Black women were more likely to have dense breasts than NH White or Hispanic women.
Are there any special considerations for screening of Black or Hispanic women?
Black women are more likely to have disease-causing variants (mutations) in BRCA. The American College of Radiology recommends Black women be evaluated in a high-risk program by age 30, even in the absence of family history [1]. Further, it is more important for Black women and Hispanic women to start screening at least by age 40 as peak breast cancer incidence occurs in the mid 40s, compared to peak in the early 50s for Caucasian women [2].
Is there a relationship between having dense breasts and pathogenic BRCA gene variants (mutations)?
Disease-causing BRCA gene variants and dense breasts are each independent risk factors for the development of breast cancer; however, pathogenic BRCA1 or BRCA2 variants are associated with a much higher risk than that of having dense breasts. As BRCA1 and BRCA2 disease-causing variants are associated with a higher risk, MRI is part of routine screening beginning at age 25 to 30 for women who have these variants, regardless of breast density. Cancers are also more likely to develop at a younger age in women with disease-causing variants in BRCA genes, and the breasts are usually dense at younger ages making mammography especially ineffective as a standalone test in such women.
What is the recommended breast screening for women with disease-causing genetic variants (mutations)?
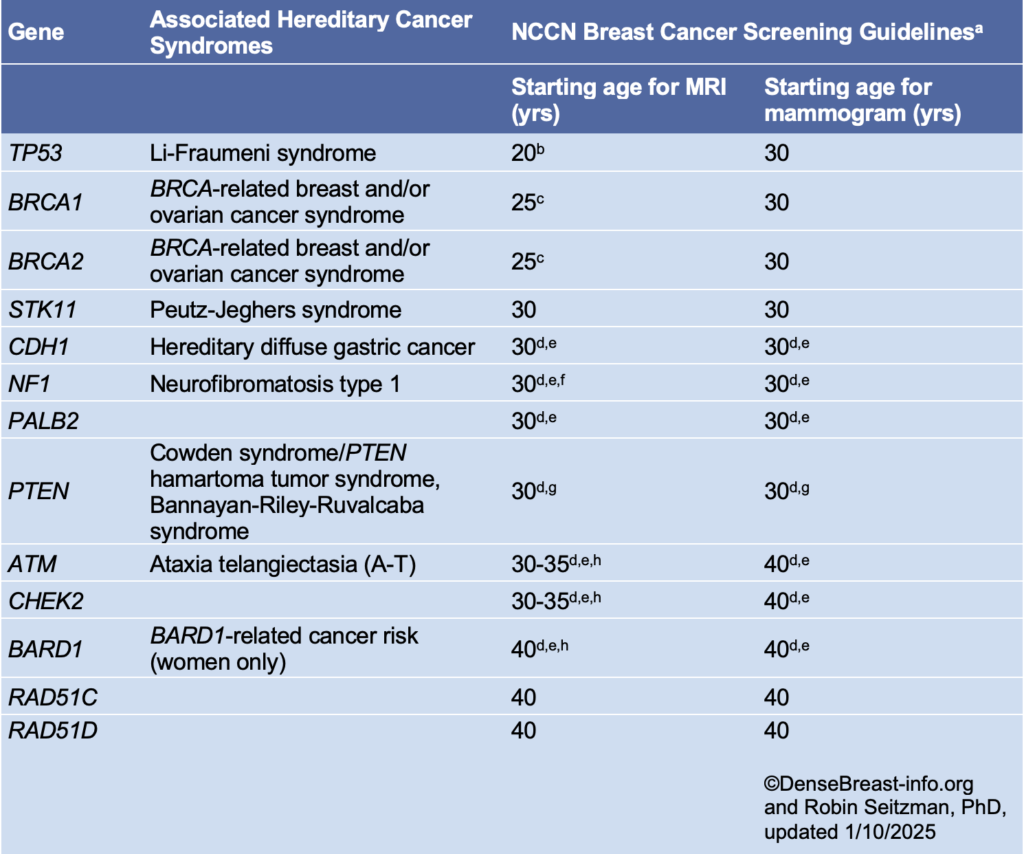
National Comprehensive Cancer Network (NCCN) guidelines recommend annual screening MRI beginning by age 25, with the addition of mammography beginning at age 30, in women who are known to carry pathogenic/likely pathogenic variants in BRCA1 or BRCA2 (unless the woman has had bilateral mastectomy), and in women who are untested first-degree relatives of those with known pathogenic/likely pathogenic variants (see table below) [1].
Women who are known to carry or are first-degree untested relatives of individuals with less common disease-causing variants [such as those associated with Li-Fraumeni syndrome (TP53); Bannayan-Riley-Ruvalcaba syndrome or Cowden syndrome (PTEN); hereditary diffuse gastric cancer (CDH1); Peutz-Jeghers syndrome (STK11); Neurofibromatosis type 1 (NF1); or Fanconi anemia (PALB2)] are also recommended for annual screening MRI, with age to start depending on the variant (see table below). Women with NF1 have elevated risk only through age 50: supplemental MRI should stop beyond age 50 for these women. For women with disease-causing variants in genes other than NF1, supplemental MRI screening should continue until age 75, after which screening should continue on an individual basis [1].
In addition to pathogenic/likely pathogenic variant carriers and their untested first-degree relatives, annual screening MRI is recommended in addition to mammography/tomosynthesis in other subgroups of women as detailed in the Dense Breasts/High-Risk Screening Guidelines.
NCCN Breast Cancer Screening Guidelines in Women Who Carry or Are First-Degree Untested Relatives of Individuals with Pathogenic Variants or Variants Known or Likely to Increase Breast Cancer Risk [1]*
*Please refer to the NCCN guideline website for updates.
a Breast awareness starting at age 18 years. Clinical breast exam every 6-12 months starting at age 25 years or 5-10 years before the earliest known breast cancer in the family. Screening should continue as indicated until age 75 (other than for women with a pathogenic NF1 mutation), after which screening should be considered on an individual basis [1].
b Start at age 20 or the age of the earliest diagnosed breast cancer in the family if younger than age 20. Adding mammography at age 30 is preferred due to the potential risk from radiation exposure in pathogenic/likely pathogenic variant carriers under age 30. Mammography can be performed from ages 20-29 years only if MRI is unavailable.
c Begin at age 25, or starting age may be individualized based on family history of a breast cancer diagnosis before at 30. From age 25-29 years, breast MRI alone is recommended. Adding mammography at age 30 is preferred due to the potential risk of radiation exposure in pathogenic/likely pathogenic variant carriers under age 30. Mammography can be performed from ages 25-29 years only if MRI is unavailable.
d For women with pathogenic/likely pathogenic variants who are treated for breast cancer and have not had bilateral mastectomy, screening should continue as described.
e Start at stated age or 5-10 years before the earliest known breast cancer in the family (whichever comes first), or based on specific pathogenic/likely pathogenic variant.
f There are currently no data to suggest an increased breast cancer risk after age 50 years in women with NF1; therefore, MRI screening may discontinue at 50 years of age in this group. In addition, the presence of breast neurofibromas may lead to false-positive MRI results; more data on sensitivity and specificity of MRI in women with NF1 are needed.
g Start at stated age or 10 years before the earliest known breast cancer in the family (whichever comes first).
h The use of MRI in these patients depends on a number of risk factors, including family history, age, breast density, and patient preference.
How do women under the age of 40 find information about breast density and their risks for breast cancer?
Dense breasts are mostly an issue affecting mammography performance so a patient generally does not need to know until they begin having mammograms. For women at normal risk, mammography is often recommended to begin at age 40. If a woman has a family history of breast cancer and has not begun mammography screening, she should speak to her doctor about personal risk factors and when mammography and possibly other screening should begin. As a general guide, if a woman’s mother or sister was diagnosed with breast cancer before age 50, she may want to begin annual screening 10 years before the relative’s age at diagnosis, but not before age 30.
European guidelines recommend double-read biennial screening digital mammography for average-risk women 50 to 69 years of age. Also encouraged are biennial screening for women 73 to 75 years of age, and annual screening for ages 40–45 to 49 [1].